DR ANTHONY MELVIN CRASTO Ph.D ( ICT, Mumbai) , INDIA 30 Yrs Exp. in the feld of Organic Chemistry. Serving chemists around the world. Helping them with websites on Chemistry.Millions of hits on google, world acclamation from industry, academia, drug authorities for websites, blogs and educational contributionn
Showing posts with label world drug tracker. Show all posts
Showing posts with label world drug tracker. Show all posts
Wednesday 25 September 2013
Toxoplasmosis: Biochemistry of Infection
 A palmitoyl thioesterase that decreases the ability of Toxoplasma gondiito invade host cells has been identified
A palmitoyl thioesterase that decreases the ability of Toxoplasma gondiito invade host cells has been identifiedFriday 20 September 2013
Molecular Storage Provides Chlorine and Phosgene Safely
Molecular Storage Provides Chlorine and Phosgene Safely
 Photodecomposition of tetrachloroethylene provides a safer source of small chlorinated building blocks for organic synthesis
Photodecomposition of tetrachloroethylene provides a safer source of small chlorinated building blocks for organic synthesisThursday 19 September 2013
Weygand/Hilgetag Preparative Organic Chemistry
Friday 6 September 2013
Fullerenols: Fulfilling our Future Energy Needs

Carbon to catalyze low-carbon, hydrogen economy
read all at
Sunday 1 September 2013
Synthesis of Tolvaptan
Synthesis of Tolvaptan
pick up all this at
YANG Chuanwei~1,MU Shuai~2,LIU Ying~3,WANG Pingbao~3,LIU Dengke~(3*)
(1.School of Pharmacy,Henan University,Kaifeng 475004;2.
School of Chemical Engineering and Technology,
Tianjin University,Tianjin 300072;3.
Tianjin Institute of Pharmaceutical Research,Tianjin 300193)
(1.School of Pharmacy,Henan University,Kaifeng 475004;2.
School of Chemical Engineering and Technology,
Tianjin University,Tianjin 300072;3.
Tianjin Institute of Pharmaceutical Research,Tianjin 300193)
Tolvaptan,a selective nonpeptide arginine vasopressin V_2 receptor antagonist,was synthesized
from 7-chloro-5-oxo-2,3,4,5-tetrahydro-1H-1-benzazepine by acylation and reduction to give 1-(4-amino-2-methylbenzoyl) -7- chloro-5-oxo-2,3,4,5-tetrahydro-1H-1-benzazepine,which was subjected to acylation with 2-methylbenzoyl chloride and reduction with sodium borohydride with an overall yield of about 45%.
from 7-chloro-5-oxo-2,3,4,5-tetrahydro-1H-1-benzazepine by acylation and reduction to give 1-(4-amino-2-methylbenzoyl) -7- chloro-5-oxo-2,3,4,5-tetrahydro-1H-1-benzazepine,which was subjected to acylation with 2-methylbenzoyl chloride and reduction with sodium borohydride with an overall yield of about 45%.
【Key Words】: tolvaptan arginine vasopressin V_2 receptor antagonist heart failure hyponatremia synthesis
【Fund】: 天津市科技计划项目(09ZCKFSH01300)
【CateGory Index】: R914
【Fund】: 天津市科技计划项目(09ZCKFSH01300)
【CateGory Index】: R914
CAJViewer7.0 supports all the CNKI file formats; AdobeReader only supports the PDF format.
| 【Citations】 | ||
| |||||
| |||||
| 【Co-citations】 | ||
| |||
| |||
Wednesday 21 August 2013
Direct synthesis of hydrogen peroxide in water in a continuous trickle bed reactor optimized to maximize productivity
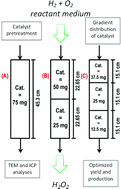
Hydrogen peroxide direct synthesis was studied in continuous mode over a 5% wt Pd/C commercial catalyst in a Trickle Bed Reactor. The target of the study was to maximize the hydrogen peroxide production. The catalyst was uniformly diluted in quartz sand at different concentrations to investigate their effects on the direct synthesis.
The amount of catalyst and the distribution of the catalyst along the bed were optimized to obtain the highest possible yield. The distribution of the catalyst along the bed gave the possibility to significantly improve the selectivity and production of hydrogen peroxide (up to 0.5% under selected conditions). Higher production rate and selectivity were found when the catalyst concentration was decreased along the bed from the top to the bottom as compared to the uniformly dispersed catalyst.
The H2/Pd ratio was found to be an important parameter that has to be investigated in the hydrogen peroxide direct synthesis. The effect of a pretreatment of the catalyst with a solution of sodium bromide and phosphoric acid was studied; the results showed how a catalyst pretreatment can lead to a real green hydrogen peroxide synthesis in water. Some optimization guidelines are also provided.
Green Chem., 2013, 15,2502-2513
DOI: 10.1039/C3GC40811F, Paper
DOI: 10.1039/C3GC40811F, Paper
*
Corresponding authors
a
Department of Chemical Engineering, Åbo Akademi University, Turku/Åbo, Finland
E-mail: bpierdom@abo.fi ;
Fax: +358 2 215 4479 ;
Tel: +358 2 215 4555
E-mail: bpierdom@abo.fi ;
Fax: +358 2 215 4479 ;
Tel: +358 2 215 4555
b
Department of Chemical Engineering and Environmental Technology, University of Valladolid, Valladolid, Spain
E-mail: jgserna@iq.uva.es
E-mail: jgserna@iq.uva.es
Iodine-mediated arylation of benzoxazoles with aldehydes
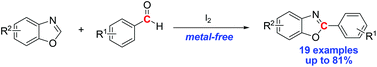
A simple and efficient methodology for the arylation of benzoxazoles with aldehydes using iodine as the mediator has been developed. The reaction proceeded smoothly with a range of substrates to give the corresponding arylated products in moderate to good yields
Green Chem., 2013, 15,2365-2368
DOI: 10.1039/C3GC41027G, Communication
DOI: 10.1039/C3GC41027G, Communication
*
Corresponding authors
a
Institute of Bioengineering and Nanotechnology, 31 Biopolis Way, The Nanos, Singapore 138669, Singapore
E-mail: ygzhang@ibn.a-star.edu.sg ;
Fax: (+65) 6478-9020
E-mail: ygzhang@ibn.a-star.edu.sg ;
Fax: (+65) 6478-9020
Friday 16 August 2013
Explosive Nanotechnology

A team of American scientists have developed highly reactive nanoenergetic formulations based on periodate salts
Multiple Metal-Free Pathways to 2-Acyloxazoles

2-Acyloxazoles can be formed from a variety of different starting materials just by tweaking the iodine oxidant
Multiple Metal-Free Pathways to 2-Acyloxazoles
Resolving Asymmetric Epoxidations

An enantioselective route to form epoxides by using kinetic resolution techniques has been developed
Wednesday 7 August 2013
A synthesis of α-amino acids via direct reductive carboxylation of imines with carbon dioxide
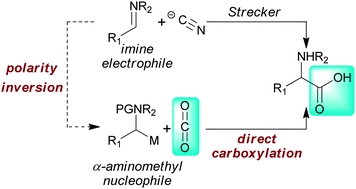
A method for the synthesis of α-amino acids by direct reductive carboxylation of aromatic imines with CO2 is described. The protocol employs readily available commercial reagents and serves as a one-step alternative to the Strecker synthesis.
A synthesis of α-amino acids via direct reductive carboxylation of imines with carbon dioxide
* Corresponding authors
a
Department of Chemistry, The Pennsylvania State University, University Park, USA
E-mail: radosevich@psu.edu ;
Tel: +1 814 867 4268
E-mail: radosevich@psu.edu ;
Tel: +1 814 867 4268
Chem. Commun., 2013,49, 5040-5042
DOI: 10.1039/C3CC42057D
Received 21 Mar 2013, Accepted 16 Apr 2013
First published online 25 Apr 2013
Tuesday 6 August 2013
Monday 5 August 2013
Homogeneous catalyst made to act more like an enzyme
| Homogeneous catalyst made to act more like an enzyme | |
 | Supramolecular encapsulation imposes electronic and steric restrictions to make catalyst more selective http://www.rsc.org/chemistryworld/2013/08/homogeneous-catalyst-supramolecular-encapsulation |
Sound solution to nanoparticle handling problems
| Sound solution to nanoparticle handling problems | |
| http://www.rsc.org/chemistryworld/2013/08/ultrasonic-technique-sticks-nanoparticles-catalytic-activity | |
 | An unexpected discovery allows scientists to stick nanoparticles together using ultrasound without destroying their catalytic activity |
Friday 2 August 2013
COLUMN CHROMATOGRAPHY

A chemist in the 1950s using column chromatography. The Erlenmeyer receptacles are on the floor.
Column chromatography in chemistry is a method used to purify individual chemical compounds from mixtures of compounds. It is often used for preparative applications on scales from micrograms up to kilograms. The main advantage of column chromatography is the relatively low cost and disposability of the stationary phase used in the process. The latter prevents cross-contamination and stationary phase degradation due to recycling.


The classical preparative chromatography column, is a glass tube with a diameter from 5 mm to 50 mm and a height of 5 cm to 1 m with a tap and some kind of a filter (a glass frit or glass wool plug – to prevent the loss of the stationary phase) at the bottom. Two methods are generally used to prepare a column: the dry method, and the wet method.
- For the dry method, the column is first filled with dry stationary phase powder, followed by the addition of mobile phase, which is flushed through the column until it is completely wet, and from this point is never allowed to run dry.
- For the wet method, a slurry is prepared of the eluent with the stationary phase powder and then carefully poured into the column. Care must be taken to avoid air bubbles. A solution of the organic material is pipetted on top of the stationary phase. This layer is usually topped with a small layer of sand or with cotton or glass wool to protect the shape of the organic layer from the velocity of newly added eluent. Eluent is slowly passed through the column to advance the organic material. Often a spherical eluent reservoir or an eluent-filled and stoppered separating funnel is put on top of the column.
The individual components are retained by the stationary phase differently and separate from each other while they are running at different speeds through the column with the eluent. At the end of the column they elute one at a time. During the entire chromatography process the eluent is collected in a series of fractions. Fractions can be collected automatically by means of fraction collectors. The productivity of chromatography can be increased by running several columns at a time. In this case multi stream collectors are used. The composition of the eluent flow can be monitored and each fraction is analyzed for dissolved compounds, e.g. by analytical chromatography, UV absorption, or fluorescence. Colored compounds (or fluorescent compounds with the aid of an UV lamp) can be seen through the glass wall as moving bands.
Overview
 Stationary phase
Stationary phase
The stationary phase or adsorbent in column chromatography is a solid. The most common stationary phase for column chromatography is silica gel, followed by alumina. Cellulosepowder has often been used in the past. Also possible are ion exchange chromatography, reversed-phase chromatography(RP), affinity chromatography or expanded bed adsorption(EBA). The stationary phases are usually finely ground powders or gels and/or are microporous for an increased surface, though in EBA a fluidized bed is used. There is an important ratio between the stationary phase weight and the dry weight of the analyte mixture that can be applied onto the column. For silica column chromatography, this ratio lies within 20:1 to 100:1, depending on how close to each other the analyte components are being eluted.
Mobile phase (eluent)
The mobile phase or eluent is either a pure solvent or a mixture of different solvents. It is chosen so that the retention factor value of the compound of interest is roughly around 0.2 - 0.3 in order to minimize the time and the amount of eluent to run the chromatography. The eluent has also been chosen so that the different compounds can be separated effectively. The eluent is optimized in small scale pretests, often using thin layer chromatography (TLC) with the same stationary phase.


There is an optimum flow rate for each particular separation. A faster flow rate of the eluent minimizes the time required to run a column and thereby minimizes diffusion, resulting in a better separation. However, the maximum flow rate is limited because a finite time is required for analyte to equilibrate between stationary phase and mobile phase, see Van Deemter's equation. A simple laboratory column runs by gravity flow. The flow rate of such a column can be increased by extending the fresh eluent filled column above the top of the stationary phase or decreased by the tap controls. Faster flow rates can be achieved by using a pump or by using compressed gas (e.g. air,nitrogen, or argon) to push the solvent through the column (flash column chromatography).
The particle size of the stationary phase is generally finer in flash column chromatography than in gravity column chromatography. For example, one of the most widely used silica gel grades in the former technique is mesh 230 – 400 (40 – 63 µm), while the latter technique typically requires mesh 70 – 230 (63 – 200 µm) silica gel.
A spreadsheet that assists in the successful development of flash columns has been developed. The spreadsheet estimates the retention volume and band volume of analytes, the fraction numbers expected to contain each analyte, and the resolution between adjacent peaks. This information allows users to select optimal parameters for preparative-scale separations before the flash column itself is attempted.


CHECK THIS VIDEO
.........................

CHECK THIS VIDEO
................................
EROS Best Reagent Award 2013
 Huw Davis, Emory University, USA, has received the the Best
Reagent Award for his widely used carbenoid precursor
Huw Davis, Emory University, USA, has received the the Best
Reagent Award for his widely used carbenoid precursorRead more
Organic Chemistry Becomes Multidisciplinary

18th European Symposium on Organic Chemistry (ESOC 2013) sees future trends in close connection to biology, physics, and materials science
Read more
Jumping Crystals

Scientists from the United Arab Emirates and Russia examined light-induced jumping crystals by kinematic analysis
Read more
Subscribe to:
Posts (Atom)

 The enzyme cyclic GMP-AMP synthase stimulates the immune response during HIV infections
The enzyme cyclic GMP-AMP synthase stimulates the immune response during HIV infections