Endo-4,7-bis(hydroxymethyl)hexahydro-1H-4,7-epoxyisoindole-1,3(2H)-dione (endo-4,7- bis(hydroxymethyl)norcantharimide), 4 (method A)
Endo-4,7-bis(hydroxymethyl)norcantharimid-5-ene (120 mg, 0.53 mmol) was dissolved in water (3 mL), Pd/C 10% was added (15 mg) and reaction mixture was placed under hydrogen atmosphere for 8 h at 24 °C. Catalyst was filtered off and washed thoroughly with water (3 × 3 mL), filtrate was evaporated under reduced pressure. Target compound 4 was obtained as white solid, yield 87% (110 mg).
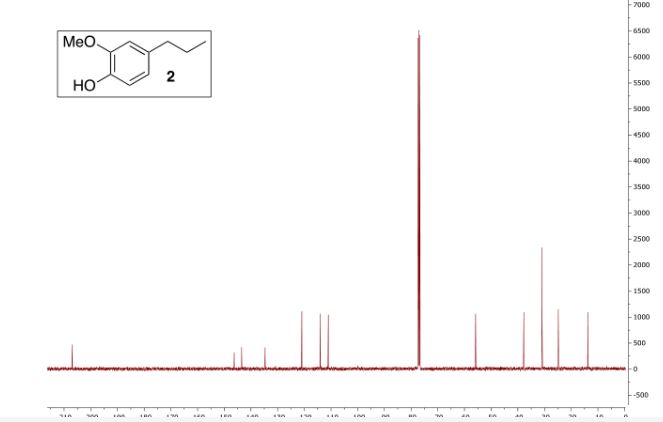
1H NMR (D2O) = 3.76 (s, 4H), 3.46 (s, 2H), 1.61-1.72 (m, 4H);
1H NMR (DMSO-d6) = 11.10 (s, 1H), 5.08 (s, 2H), 3.66 (s, 4H), 3.37 (s, 2H), 1.71 (m, 2H), 1.49 (m, 2H);
13C NMR (D2O) = 179.0, 88.8, 60.7, 52.3, 27.0 ppm.
m/z HRMS (ESI) Calcd. for C10H13NO5 [M+Na]: 250.0686. Found 250.0696.
Green Chem., 2017, Advance Article
DOI: 10.1039/C7GC02211E, Paper
F. A. Kucherov, K. I. Galkin, E. G. Gordeev, V. P. Ananikov
Efficient one-pot synthesis of tricyclic compounds from biobased 5-hydroxymethylfurfural (HMF) is described using a [4 + 2] cycloaddition reaction.
Efficient route for the construction of polycyclic systems from bioderived HMF
Author affiliations
*Corresponding authors
aZelinsky Institute of Organic Chemistry, Russian Academy of Sciences, Leninsky pr. 47, Moscow 119991, Russia
E-mail: val@ioc.ac.ru
Web: http://AnanikovLab.ru
//////////