http://pubs.rsc.org/en/Content/ArticleLanding/2016/GC/C6GC00385K?utm_source=feedburner&utm_medium=feed&utm_campaign=Feed%3A+rss%2FGC+%28RSC+-+Green+Chem.+latest+articles%29#!divAbstract
Despite the undisputed advances and progress in metal-catalyzed C–H functionalizations, this atom-economical approach had thus far largely been developed with the aid of various metal catalysts that were operative in a homogeneous fashion.
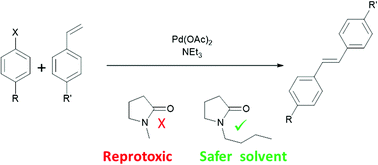
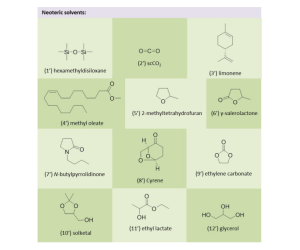
While thereby major progress was accomplished, these catalytic systems featured notable disadvantages, such as low catalyst recyclability. This review summarizes the development of user-friendly, recyclable and easily separable heterogeneous catalysts for C–H activation.
This strategy was characterized by a remarkably broad substrate scope, considerable levels of chemo- and site-selectivities and proved applicable to C–C as well as C–heteroatom formation processes.
Thus, recyclable catalysts were established for arylations, hydroarylations, alkenylations, acylations, nitrogenations, oxygenations, or halogenations, among others. The rapid recent progress in selective heterogeneous C–H functionalizations during the last decade until December 2015 is reviewed.
Heterogeneous catalytic approaches in C–H activation reactions
*
Corresponding authors
a
Laboratory of Green Synthetic Organic Chemistry, Dipartimento di Chimica Biologia e Biotecnologie, Università di Perugia, Via Elce di Sotto, 8 – 06123 Perugia, Italy
E-mail: luigi.vaccaro@unipg.it
Web: http://www.dcbb.unipg.it/greensoc
E-mail: luigi.vaccaro@unipg.it
Web: http://www.dcbb.unipg.it/greensoc
b
Institut für Organische und Biomolekulare Chemie, Georg-August-Universität Göttingen, Tammannstrasse 2, 37077 Göttingen, Germany
E-mail: Lutz.Ackermann@chemie.uni-goettingen.de
Web: http://www.ackermann.chemie.uni-goettingen.de
E-mail: Lutz.Ackermann@chemie.uni-goettingen.de
Web: http://www.ackermann.chemie.uni-goettingen.de
Green Chem., 2016, Advance Article
DOI: 10.1039/C6GC00385K
Laboratory
of Green Synthetic Organic Chemistry, Dipartimento di Chimica Biologia e
Biotecnologie, Università di Perugia, Via Elce di Sotto, 8 – 06123
Perugia, Italy
E-mail: luigi.vaccaro@unipg.it
Web: http://www.dcbb.unipg.it/greensoc
E-mail: luigi.vaccaro@unipg.it
Web: http://www.dcbb.unipg.it/greensoc
Extra clips

C-H Activation :: Wiley-VCH Hot Topics
The Yu Lab
www.scripps.edu
"Ligand-Enabled
Triple C-H Activation Reactions: One-Pot Synthesis of Diverse
4-Aryl-2-quinolinones from Propionamides" Angew. Chem. Int. Ed. 2014,
53, ...
////////////