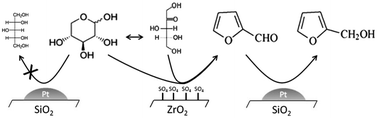
7,7-dichlorobicyclo[4.1.0]heptane (7,7-dichloronorcarane
 +
+
| NaOH,Pr3N |
 |
![7,7-Dichlorobicyclo[4.1.0]heptane 7,7-Dichlorobicyclo[4.1.0]heptane](http://kriemhild.uft.uni-bremen.de/nop/img/chents/small/91_en.gif) +
+
Synthesis of 7,7-dichlorobicyclo[4.1.0]heptane (7,7-dichloronorcarane) from cyclohexene |
| Reaction type: | addition to alkenes, elimination, cycloaddition |
| Substance classes: | alkene, carbene, chloroalkane |
| Techniques: | stirring with magnetic stir bar, adding dropwise with an addition funnel, distilling under reduced pressure, evaporating with rotary evaporator, shaking out, extracting, filtering, use of an ice cooling bath, heating with oil bath |
Instruction (batch scale 100 mmol)
Equipment
100 mL three neck round bottom flask, reflux condenser, addition funnel with pressure
balance, heatable magnetic stirrer, magnetic stir bar, thermometer for inside of the flask,
separating funnel, destillation apparatus, rotary evaporator, oil bath, ice bath, vacuum pump
Substances
cyclohexene (bp 83 °C) 8.21 g (10.1 mL, 100 mmol)
chloroform (bp 61 °C) 48.0 g (32.7 mL, 400 mmol)
sodium hydroxide 16.0 g (400 mmol)
tri-n-propylamine (bp 156 °C) 0.14 g (0.19 mL, 1.0 mmol)
water 16 mL
ethanol (bp 78 °C) 1 mL
n-pentane (bp 36 °C) 120 mL
sodium sulfate for drying about 5 g
sodium chloride about 18 g
Reaction
Into a 100 mL three neck round bottom flask equipped with a reflux condenser addition
funnel, thermometer for measuring the inside temperature and magnetic stir bar, 8.21 g
(10.1 mL, 100 mmol) cyclohexene 0.14 g (0.19 mL, 1.0 mmol) tri-n-propylamine, 48.0 g
(32.7 mL, 400 mmol) chloroform and 1 mL ethanol is added. The mixture is cooled to 0 °C
with an ice bath, then under stirring and further cooling in the ice bath a solution of 16.0 g
(400 mmol) sodium hydroxide in 16 mL water is added through an addition funnel. The
mixture should be stirred vigourously during the next 20 minutes at 0 °C. After this time the
mixture is further stirred during 1 hour at room temperature and 3 hours at 50 °C.
Work up
Chloroform is evaporated with a rotary evaporator, then the residue is transferred with about
50 mL water and 30 mL n-pentane into a separating funnel. The organic phase is separated,
the aqueous phase is further extracted three times with 30 mL pentane. If an emulsion is
formed the aqueous phase is saturated with NaCl. The combined organic phases are dried over
sodium sulfate. The solution is filtered from sodium sulfate and the solvent is evaporated with
a rotary evaporator, yielding a nearly colourless liquid as crude product. The crude yield is
14.6 g. The crude product is distilled under reduced pressure.
Yield: 13.6 g (82.3 mmol, 82%), colourless liquid; bp 77 °C (11 hPa)
Equipment
100 mL three neck round bottom flask, reflux condenser, addition funnel with pressure
balance, heatable magnetic stirrer, magnetic stir bar, thermometer for inside of the flask,
separating funnel, destillation apparatus, rotary evaporator, oil bath, ice bath, vacuum pump
Substances
cyclohexene (bp 83 °C) 8.21 g (10.1 mL, 100 mmol)
chloroform (bp 61 °C) 48.0 g (32.7 mL, 400 mmol)
sodium hydroxide 16.0 g (400 mmol)
tri-n-propylamine (bp 156 °C) 0.14 g (0.19 mL, 1.0 mmol)
water 16 mL
ethanol (bp 78 °C) 1 mL
n-pentane (bp 36 °C) 120 mL
sodium sulfate for drying about 5 g
sodium chloride about 18 g
Reaction
Into a 100 mL three neck round bottom flask equipped with a reflux condenser addition
funnel, thermometer for measuring the inside temperature and magnetic stir bar, 8.21 g
(10.1 mL, 100 mmol) cyclohexene 0.14 g (0.19 mL, 1.0 mmol) tri-n-propylamine, 48.0 g
(32.7 mL, 400 mmol) chloroform and 1 mL ethanol is added. The mixture is cooled to 0 °C
with an ice bath, then under stirring and further cooling in the ice bath a solution of 16.0 g
(400 mmol) sodium hydroxide in 16 mL water is added through an addition funnel. The
mixture should be stirred vigourously during the next 20 minutes at 0 °C. After this time the
mixture is further stirred during 1 hour at room temperature and 3 hours at 50 °C.
Work up
Chloroform is evaporated with a rotary evaporator, then the residue is transferred with about
50 mL water and 30 mL n-pentane into a separating funnel. The organic phase is separated,
the aqueous phase is further extracted three times with 30 mL pentane. If an emulsion is
formed the aqueous phase is saturated with NaCl. The combined organic phases are dried over
sodium sulfate. The solution is filtered from sodium sulfate and the solvent is evaporated with
a rotary evaporator, yielding a nearly colourless liquid as crude product. The crude yield is
14.6 g. The crude product is distilled under reduced pressure.
Yield: 13.6 g (82.3 mmol, 82%), colourless liquid; bp 77 °C (11 hPa)
Operating scheme
 Operating scheme
Operating schemeEquipment |

GC: pure productcolumnSE-54, L= 25 m, ID 0.32 mm, DF 0.25 µm (Macherey & Nagel)inletGerstel KAS, injector: 250°C, split injection 1:20, 0.15 µLcarrier gasN2, pre-column pressure 62 kPa, 1.04 mL/minoven80 °C (1 min), 10 °C/min --> 250 °C (30 min)detectorFID, 275 °Cintegrationpercent concentration calculated from relative peak area

| 1H-NMR: 7,7-Dichlorobicyclo[4.1.0]heptane | |||
| 500 MHz, CDCl3 | |||
| delta [ppm] | mult. | atoms | assignment |
| 1.12-1.36 | m | 4 H | C4-H, C5-H |
| 1.61-1.71 | m | 4 H | C3-H, C6-H |
| 1.88-1.98 | m | 2 H | C1-H, C2-H |
| 7.26 | s | 1 H | CHCl3 |

| 3C-NMR: 7,7-Dichlorobicyclo[4.1.0]heptane | |||
| 125 MHz, CDCl3 | |||
| delta [ppm] | assignment | ||
| 18.9 | C4, C5 | ||
| 20.2 | C3, C6 | ||
| 25.8 | C2, C1 | ||
| 67.4 | C7 | ||
| 76.5-77.5 | CDCl3 | ||

| IR: 7,7-Dichlorobicyclo[4.1.0]heptane | |||
| [Film, T%, cm-1] | |||
| [cm-1] | assignment | ||
| 2944, 2855 | aliph. C-H valence | ||
| 796 | C-Cl valence | ||
ORGANIC SPECTROSCOPY





















 amcrasto@gmail.com
amcrasto@gmail.com