In a ground-breaking feat of protein engineering, US researchers have designed a synthetic enzyme that catalyses the Diels-Alder reaction - something that has not been seen in nature. The reaction is key to many organic syntheses and suggests that artificial enzymes could soon become part of the synthetic chemist's toolkit.
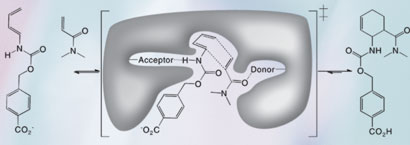
The Diels-Alder reaction - diene and dienophile undergo a pericyclic [4 + 2] cycloaddition to form a chiral cyclohexene ring. The image also shows the design target active site, with hydrogen bond acceptor and donor groups activating the diene and dienophile and a complementary binding pocket holding the two substrates in an orientation optimal for catalysis
Science 16 July 2010:
Vol. 329 no. 5989 pp. 309-313 DOI: 10.1126/science.1190239 |

No comments:
Post a Comment