Anurag Mandalika and Troy Runge
DOI: 10.1039/C2GC35759C, Paper
http://pubs.rsc.org/en/content/articlelanding/2012/gc/c2gc35759c
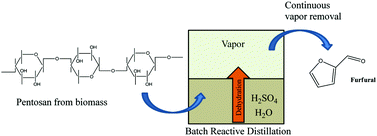
Batch reactive distillation of biomass hydrolysates was employed to produce furfural in high yield using an entirely aqueous acid-catalyzed process.
Dilute aqueous solutions of furfural were produced in high yield from biomass hydrolysates using an acid-catalyzed batch reactive distillation process that separated the vapor phase from the aqueous reactant medium. Hot water hydrolysates from hybrid poplar, miscanthus, switchgrass and corn stover were dehydrated using sulfuric acid. The vapor fraction from the reactor was condensed to produce furfural in excess of 85% of the theoretical yield based on total pentose. Using xylose as the model compound, and temperature and acid concentration as the variables, the process conditions were optimized by the construction of a three-level statistical model. Hot water hydrolysis of biomass provided with a cellulose-rich solid fraction which has potential for conversion into pulp or cellulosic ethanol, while the liquid fraction, rich in hemicellulose sugars, was converted into furfural. Fractionating the biomass allows for exploration of the concept of the integrated biorefinery where the hemicellulose sugars are not underutilized or encountered as potential inhibitors during microbial conversions of the solid stream, but are converted into furfural, a valuable chemical precursor. The availability of the cellulose fraction for further conversion into pulp or ethanol gives the current process a major advantage over the conventional batch process used in industry, where theoretical yields do not exceed 45–50% with the conspicuous absence of a usable cellulose stream.
Thanks to vast readership for million hits on google on all my sites, you can be out of storm safely to shore if you have will power and determination, help is an email away, amcrasto@gmail.com check out http://amcrasto.bravesites.com/





